 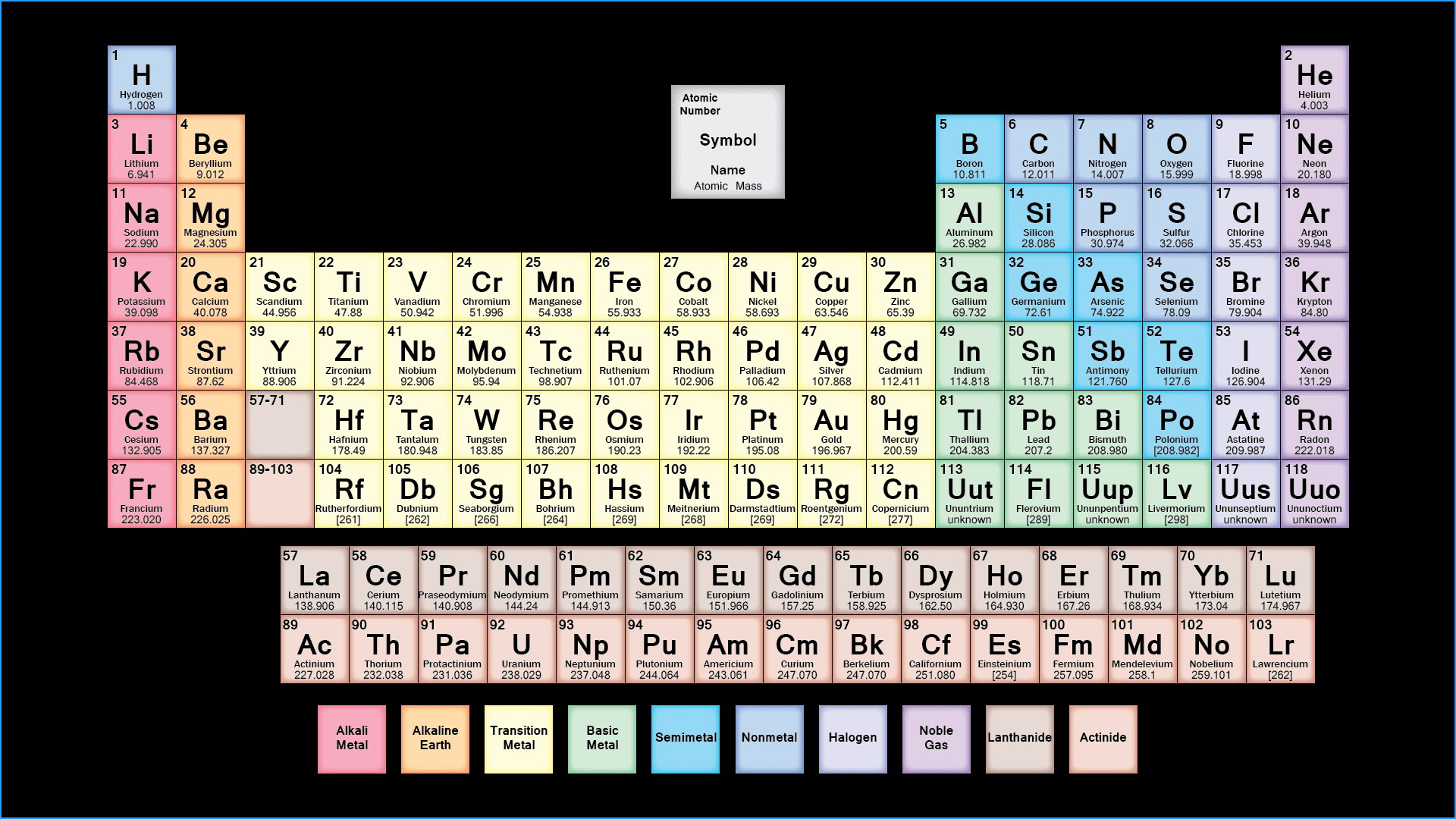
Elements of this block are also called inner transition elements.Ĭheck the Development of the Periodic Table here.Electronic configuration of lanthanides is 6s25d0-24f1-14.The electronic configuration of actinides is irregular.The f-block consists of two series lanthanides and actinides of the periodic table.The three series of transition metals are known 3d series, 4d series and 5d series.This block contains the elements of groups 3 to 12 of the periodic table.Valence shell electronic configuration is (n-1)d1-10 ns1-2.In d-block elements, a valence electron enters in d-orbital.These elements are called transition elements.At the end of each period is a noble gas element with a closed valence shell ns2np6 configuration.In fact, the heaviest element in each p-block group is the most metallic in nature. The non-metallic character of elements decreases down the group.It is interesting to note that the non-metals and metalloids exist only in the p-block of the periodic table.The outer electronic configuration of these elements is ns2 np1.The p-Block Elements comprise those belonging to Group 13 to 18 and these together with the s-Block Elements are called the Representative Elements or Main Group Elements.The compounds of the s-block elements, with the exception of those of lithium.Because of high reactivity, they are never found pure in nature.These elements are soft metals, electropositive and form basic oxides.They are all reactive metals with low ionization enthalpies.The elements of Group 1 ( alkali metals) and Group 2 ( alkaline earth metals) which have ns1 and ns2 outermost electronic configuration belong to the s-Block Elements.In these elements, the valence electron enters an s-orbital.s-block, p-block, d-block, f-block Elementsĭepending upon the type of orbital receiving the valence electrons, the elements can be classified into four following blocks: Revised Periodic Table of the Elements (2011) by D.E.Read more about the Position of Hydrogen in Periodic Table, here.An Alternate Graphical Representation of PEriodic Table of Chemical Elements by Mohd Abubakr (PDF – 8 pages).The Periodic Table of the Elements – in Pictures by Sercon (PDF).Mendeleev and the Periodic Table by Oak Park – Unified School District (PDF – 12 pages).The Many Looks of the Periodic Table by Gary Katz (PDF – 3 pages).Causal Explanation and the Periodic Table by Lauren N.The Periodic Table of Elements by Jefferson Lab (PDF).Periodic Table – Atomic Properties of the Elements by National Institute of Standards and Technology (PDF).Periodic Table of Elements by The University of Sydney (PDF).Is the Periodic Table all right? by EPJ Web of Conferences (PDF).Periodic Table of the Elements by Michigan State University (PDF).Periodic Table of Elements by IUPAC (PDF).The Periodic Table of Elements by Widener University (PDF). 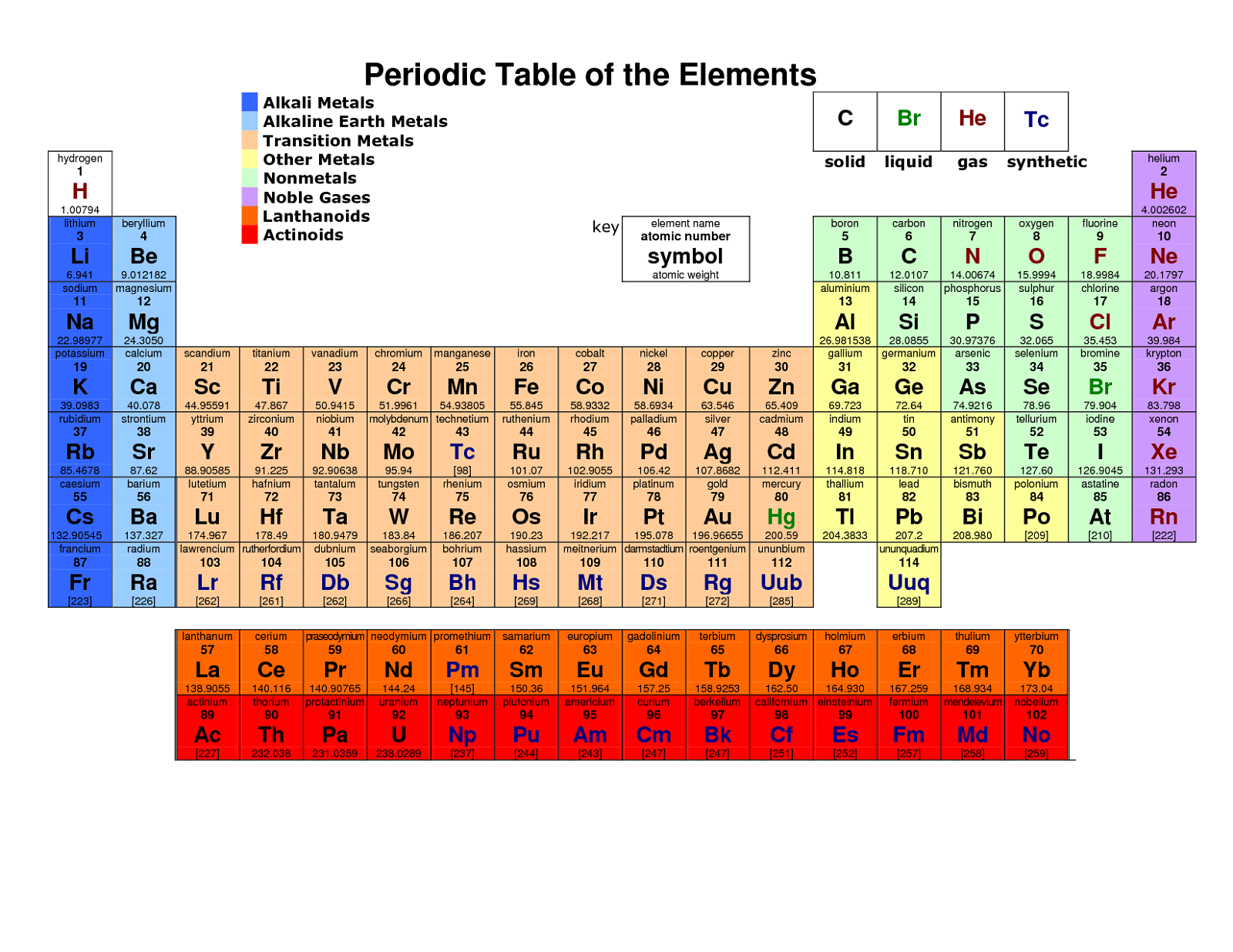
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


 RSS Feed
RSS Feed
